Caroline Biojone - Translational Psychopharmacology
Research Focus
Our group investigates novel molecular mechanisms regulating neural plasticity and develops new compounds for the pharmacological modulation of these targets.
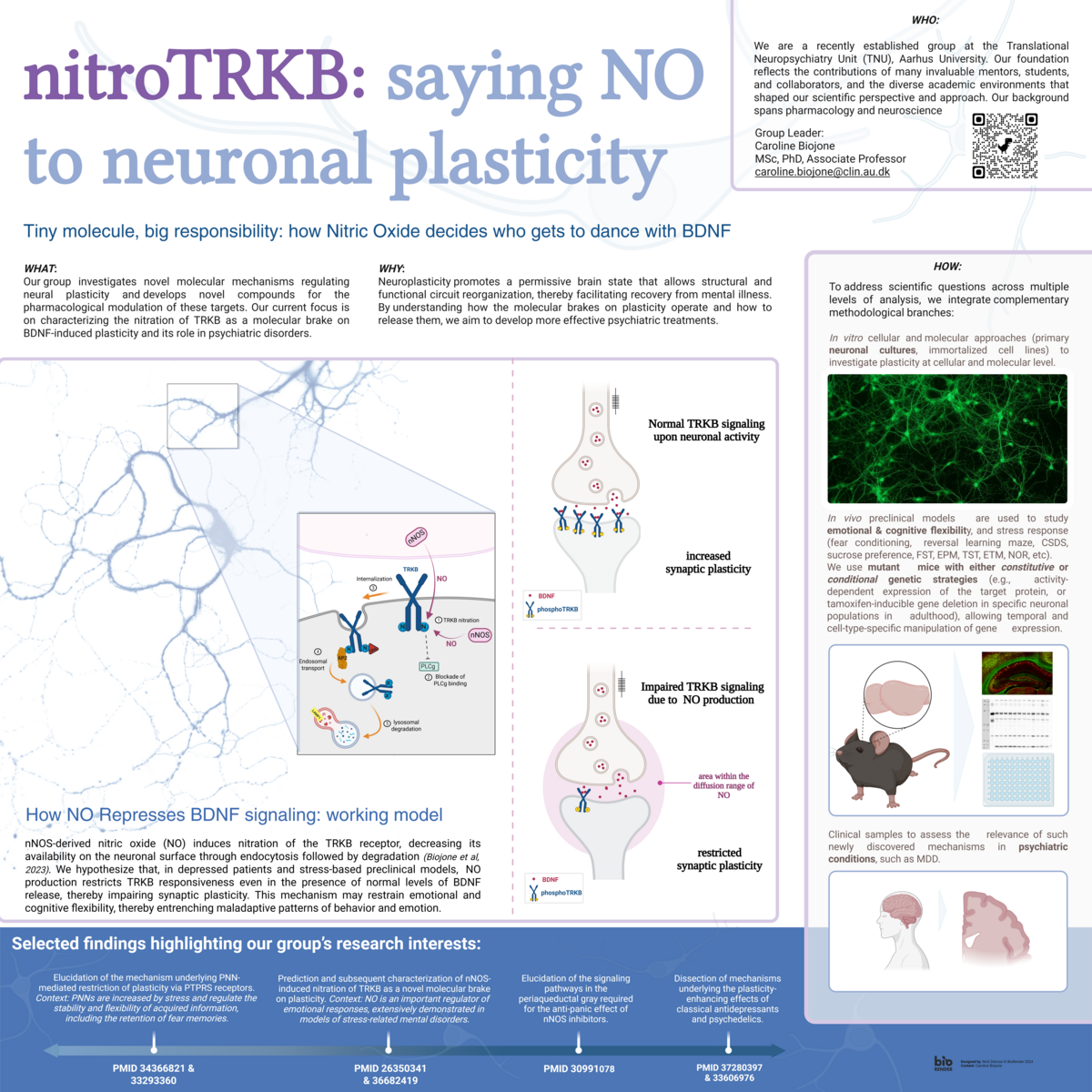
One of our main projects focuses on understanding how nitric oxide (NO) and the BDNF–TRKB system interact to refine neural networks under both physiological and pathological conditions (see figure below).
Neuroplasticity promotes a permissive brain state that enables structural and functional circuit reorganization, thereby facilitating recovery from mental illness.
By uncovering how the molecular “brakes” on plasticity operate, and how they can be released, we aim to develop more effective treatments for psychiatric disorders.
Experimental Strategy
To address our scientific questions across multiple levels of analysis, we integrate four complementary methodological branches:
- In silico modeling and prediction
- In vitro cellular and molecular approaches (primary neuronal cultures, cell lines)
- In vivo preclinical models to study emotional and cognitive flexibility (e.g., fear conditioning, reversal learning, chronic social defeat stress, sucrose preference).
We employ mutant mice with constitutive or conditional genetic strategies (e.g., activity-dependent expression of target proteins, or tamoxifen-inducible gene deletion in specific neuronal populations during adulthood), allowing temporal and cell-type-specific manipulation of gene expression.
- Clinical samples to assess the relevance of newly discovered mechanisms in psychiatric conditions
Selected Findings Illustrating Our Research Focus
- PNN-mediated restriction of plasticity via PTPRS receptors (1,2).
Context: Perineuronal nets (PNNs), which are increased by stress, regulate the stability and flexibility of acquired information, including fear memory retention.
- nNOS-induced nitration of TRKB as a novel molecular brake on plasticity (3,4).
Context: Nitric oxide (NO) is a key regulator of emotional responses, as demonstrated in models of stress-related mental disorders.
- Signaling pathways in the periaqueductal gray underlying the anti-panic effects of nNOS inhibitors (5).
Mechanisms underlying the plasticity-enhancing effects of classical antidepressants, S-ketamine, and psychedelics (6,7).
References
1. Lesnikova et al. J. Neurosci. 2020
2. Lesnikova et al. Front. Synaptic Neurosci. 2021
3. Biojone et al. CNS Neurol. Disord. Drug Targets 2015
4. Biojone et al. Prog. Neurobiol. 2023
5. Ribeiro et al. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019
6. Casarotto et al. Cell 2021
7. Moliner et al. Nat. Neurosci. 2023